|
Note that in Fe(NO 3) 3, there are three nitrate ions, i.e., one iron atom, three nitrogen atoms, and nine oxygen atoms, in the formula unit of the compound. 9.1.2 Define a polyatomic ion and write the names and formulas of. For example, iron(III) nitrate is Fe(NO 3) 3 sodium carbonate is Na 2CO 3 ammonium phosphate is (NH 4) 3PO 4 potassium permanganate is KMnO 4 and calcium phosphate is Ca 3(PO 4) 2. 1 Identify the charges on mona- tomic ions by using the peri- odic table, and name the ions. You will always be allowed a periodic table, which makes. Just like the subscript to the right of the monoatomic anion tells how many atoms of the anions are there, the subscript to the right of the small bracket around a polyatomic anion tells how many polyatomic anions are there in the compound. A mastery of the common ions, their formulas, and their charges is essential to success in chemistry. Any such molecule that has nonzero net charge is a polyatomic ion. The carbonate ion consists of one carbon atom and three oxygen atoms, and carries an overall charge of 2 2. Together, they comprise a single ion with a 1+ 1 + charge and a formula of NH+4 NH 4 +. The ammonium ion consists of one nitrogen atom and four hydrogen atoms. If a subscript is needed for the anion, place the polyatomic ion within small brackets and write the subscript outside the bracket. Polyatomic ions are molecular ions composed of two or more atoms bonded by covalent bonds and acting as a single unit, but unlike molecules, they have a net charge on them. protons electrons So, hydroxide has a net charge of 1 because its number of electrons exceeds its number of protons by one. A polyatomic ion is an ion composed of more than one atom. The writing formula of compounds containing polyatomic ions is the same as writing the formula of a binary ionic compound, except that the polyatomic ions must remain intact as a unit. The polyatomic ion acts as a single unit, i.e., they are molecular ions. Writing formulae of compounds containing polyatomic ions 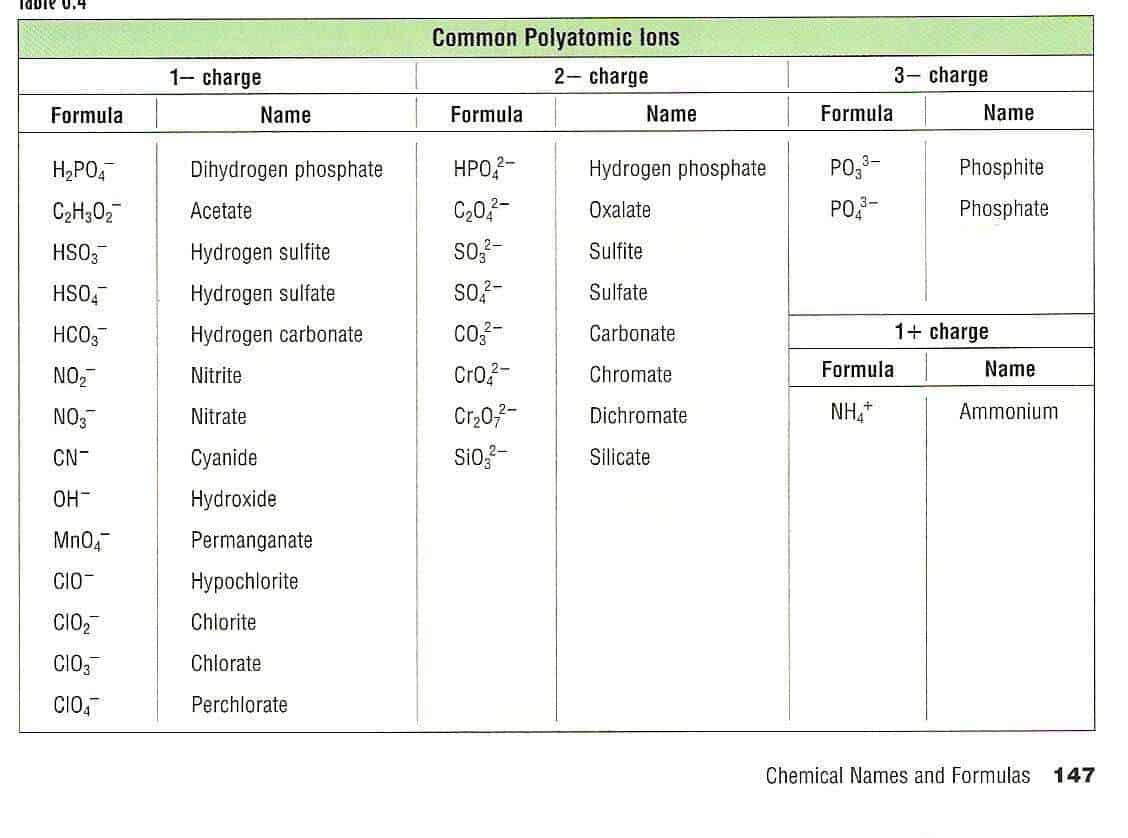 
Table 1: Names of some of the common polyatomic ions Formula Table 3.3.1 lists the formulas and names of some of the common polyatomic ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
